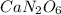Answer:
Step-by-step explanation:
When calculating an empirical formula from percentages, assume you have a 100g sample. This allows you to convert the percentages directly to grams, because X % of 100g is X grams.
So:
24.42 % = 24.42 g Ca, 17.07% = 17.07g N, 58.5% = 58.5g O
The next step is to divide each mass by their molar mass to convert your grams to moles.
24.42/40.08 = 0.6092 mol
17.07/14.01 = 1.218 mol
58.85/15.99 = 3.680 mol
Then you will divide all of your mol values by the SMALLEST number of moles. This gives you whole numbers that are the mole ratio (subcripts) of the empircal formula.
0.6092 mol/0.6092 mol = 1
1.218 mol/0.6092 mol = 2
3.680 mol/0.6092 mol = 6
So the empirical formula is