The given question is incomplete. The complete question is:
A gas has a volume of 590 ml at a temperature of
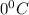 .What volume will the gas occupy at
.What volume will the gas occupy at
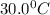 . Which gas law is this?
. Which gas law is this?
Answer: 655 ml , Charle's Law
Step-by-step explanation:
To calculate the final volume of the system, we use the equation given by Charles' Law. This law states that volume of the gas is directly proportional to the temperature of the gas at constant pressure.
Mathematically,
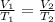
where,
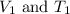 are the initial volume and temperature of the gas.
are the initial volume and temperature of the gas.
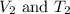 are the final volume and temperature of the gas.
are the final volume and temperature of the gas.
We are given:
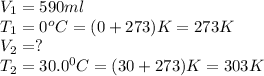
Putting values in above equation, we get:
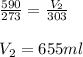
Thus the volume at
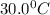 is 655 ml
is 655 ml