Answer:
The pressure of the gas at 23 C is 179.92 kPa.
Step-by-step explanation:
Gay-Lussac's law indicates that, as long as the volume of the container containing the gas is constant, as the temperature increases, the gas molecules move faster. Then the number of collisions with the walls increases, that is, the pressure increases. That is, the pressure of the gas is directly proportional to its temperature.
In short, when there is a constant volume, as the temperature increases, the pressure of the gas increases. And when the temperature is decreased, the pressure of the gas decreases.
Gay-Lussac's law can be expressed mathematically as follows:
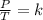
Studying two states, one initial 1 and the other final 2, it is satisfied:
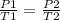
In this case:
- P1= 310 kPa
- T1= 237 C= 510 K (being 0 C= 273 K)
- P2= ?
- T2= 23 C= 296 K
Replacing:
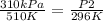
Solving:
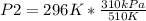
P2= 179.92 kPa
The pressure of the gas at 23 C is 179.92 kPa.