To find the number of atoms contained in 1.52 moles of stronium, we will use Avogadro's number. This number tells us that in one mole of any substance, there are 6.022x10^23 molecules, atoms or particles. Therefore, the number of atoms will be:
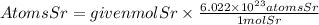
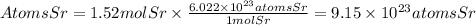
Answer: In 1.52 moles of strontium there are 9.15x10^23atoms