In order to find the volume of glycerol the student should pour out, we need to know the relation between its mass M, its volume V, and its density d:
V = M/d
Now, observe that:
M = 50.0 g
d = 1.26 g * cm-³ (this means each cubic centimeter of the substance contains 1.26 g)
Then, we can use those values in the formula for the volume:
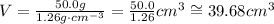
Notice that the number of significant figures of each given measure is 3 (the zero after the dot in 50.0 also counts as a significant figure).
Therefore, the answer with the correct number of significant figures is:
39.7 cm³