For the (B) the element that as this electronic configuration is Krypton. The easiest way to do this is to add all the numbers that are associated with the orbitals.
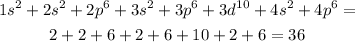
When you do this method and count up all the electrons to determine the element you would realise that option C Neon(1s2 2s2 2p6) is in the group on the periodic table. Similarly, the last orbital tells the group in which the element belongs so this can be used as well. We can see that the element in question and option C last orbital is p6.
C) 1s2 2s2 2p3 =1s2 2s2 2p6 3s2 3p3