Answer:
The amount of Au-198 remaining after 5days is 0.554mg
Step-by-step explanation:
Given:
initial amount of Au-198= 2mg
half-life = 2.7 days
To find:
Amount remaining after 5 days
To determine the amount remaining, we will apply the half-life formula:
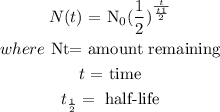
We will write the equation in terms of t. Substitute other values into the formula except the value of t:
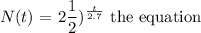
To get the amount remaining after 5 days, we will substitute t with 5
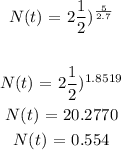
The amount of Au-198 remaining after 5days is 0.554mg