The question provided the equation for the decomposition of PH3 and the rate of decomposition for this compound (0.0605 s^-1), and requires us to calculate the final concentration of PH3 after 16.00 s given that the reaction started with 0.95 M of this compound.
The following information was provided by the question:
- Balanced chemical equation:
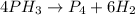
- rate of decomposition of PH3 = 0.0605 1/s
- initial concentration of PH3 = 0.95 M
- time of reaction = 16.00 s
The rate of the reaction given can be written as:
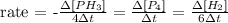
From the expression of rate above and knowing the rate of decomposition of PH3, we can calculate the Δ[PH3] and, using the initial concentration given, we can find the final concentration of PH3 after 16.00s.
First, let's calculate the Δ[PH3]: