The given question is incomplete. The complete question is:
When 136 g of glycine are dissolved in 950 g of a certain mystery liquid X, the freezing point of the solution is 8.2C lower than the freezing point of pure X. On the other hand, when 136 g of sodium chloride are dissolved in the same mass of X, the freezing point of the solution is 20.0C lower than the freezing point of pure X. Calculate the van't Hoff factor for sodium chloride in X.
Answer: The vant hoff factor for sodium chloride in X is 1.9
Step-by-step explanation:
Depression in freezing point is given by:
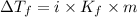
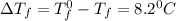 = Depression in freezing point
= Depression in freezing point
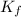 = freezing point constant
= freezing point constant
i = vant hoff factor = 1 ( for non electrolyte)
m= molality =
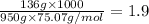
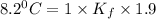
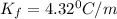
Now Depression in freezing point for sodium chloride is given by:
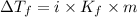
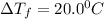 = Depression in freezing point
= Depression in freezing point
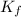 = freezing point constant
= freezing point constant
m= molality =
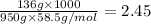
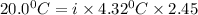
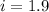
Thus vant hoff factor for sodium chloride in X is 1.9