Answer: The correct option is enough solvent to make 1.00 L of solution
Step-by-step explanation:
A solution consists of solute and solvent. A solute is defined as the component present in a smaller proportion while the solvent is defined as the component that is present in a larger proportion.
Molarity is defined as the amount of solute expressed in the number of moles present per liter of solution. The units of molarity are mol/L. The formula used to calculate molarity:
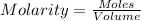 .......(1)
.......(1)
We are given:
Molarity of solution = 0.500 M
Moles of solute = 0.500 moles
Putting values in equation 1, we get:
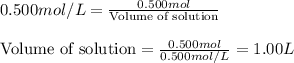
Hence, the correct option is enough solvent to make 1.00 L of solution