Answer: 0.45 moles of
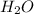 will be produced from 0.15 moles of propanol.
will be produced from 0.15 moles of propanol.
Step-by-step explanation:
The balanced chemical reaction is:
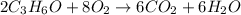
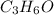 is the limiting reagent as it limits the formation of product and
is the limiting reagent as it limits the formation of product and
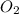 is the excess reagent.
is the excess reagent.
According to stoichiometry :
2 moles of
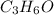 produces = 6 moles of
produces = 6 moles of
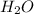
Thus 0.15 moles of
 will produce=
will produce=
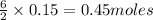 of
of
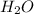
Thus 0.45 moles of
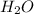 will be produced from 0.15 moles of propanol.
will be produced from 0.15 moles of propanol.