Answer:

Step-by-step explanation:
Density is found by dividing the mass by the volume.
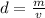
The mass of the object is 11 grams and the volume is 13 milliliters.
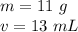
Substitute the values into the formula.
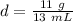
Divide.
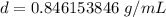
Round to the nearest hundredth. The 6 in the thousandth place tells us to round the 4 to a 5.
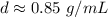
The density is about 0.85 grams per milliliter.