Final Answer:
The energy levels of an electron in a hydrogen atom, according to the Bohr model, we can find the energy change (ΔE ). the energy (in J) of an electron in the n=4 level of the Bohr hydrogen atom is 7.56944444 × 10⁻²⁰ J. The answer not provided in the option.
Step-by-step explanation:
The energy levels of an electron in a hydrogen atom, according to the Bohr model, are given by the formula:
Eₙ =
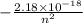
where n is the principal quantum number.
For the transition from n = 6 to n = 4, we can find the energy change (ΔE ) by subtracting the energy at n = 4 from the energy at n = 6:
ΔE =
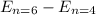
ΔE =
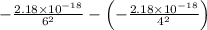
Now, calculate this expression to find ΔE.
ΔE =
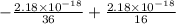
ΔE = -0.0605555556 × 10⁻¹⁸ + 0.13625 × 10⁻¹⁸
ΔE = 0.0756944444 × 10⁻¹⁸
ΔE = 7.56944444 × 10⁻²⁰
So, the energy (in J) of an electron in the n=4 level of the Bohr hydrogen atom is 7.56944444 × 10⁻²⁰ J.