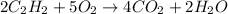A complete combustion reaction like the one you mention is a reaction that will produce carbon dioxide and water using oxygen as the oxidizing agent. The fuel can vary and from there different numbers of CO2 and water will be obtained. Let us first write what the reaction would be taking into account the unbalanced reactants and products:
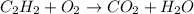
Now, to balance the equation we must count the number of atoms of each element on each side of the reaction.
In the previous scheme, we see that neither the carbon nor the oxygen are balanced, we can start by balancing the carbon. We have two carbon atoms in the reactants, so we put the coefficient two in front of the CO2 molecule.
I will be updating the scheme as I describe the procedure so that you are aware of it.
Now we must balance the oxygen, we have 5 atoms in the products and two in the reactants. To balance it, since we have an odd number in the products, we must adjust it to an even number. We place coefficient 2 in front of the H2O molecule. We have 6 oxygen atoms in the products, to have 6 oxygen atoms in the reactants we must place the coefficient 3 in front of the O2 molecule.
Now we have 6 oxygen atoms on each side of the reaction, we continue now with hydrogen. We have 4 hydrogens in the reactants, so we put the coefficient 2 in front of the C2H2 molecule.
Now the hydrogen is balanced, but the number of carbons changed and we must balance them again. We place the coefficient 4 in the CO2 molecule, so we will have 4 carbons on each side of the reaction.
In doing so, the number of oxygens was modified, so to finally have a balanced reaction we put the coefficient 5 in the O2 molecule to have 10 oxygen atoms on each side.
So, the balanced reaction will be: