Answer: 2.50 x 10^23 atoms of titanium correspond to 0.415 moles of titanium.
Step-by-step explanation:
The question requires us to calculate the number of moles of titanium (Ti) that correponds to 2.50 x 10^23 atoms of Ti.
We can apply the Avogadro's number to solve this problem: according to this proportionality constant, there are 6.022 x 10^23 particles in 1 mol of any compound (particles can be atoms, ions, molecules etc).
Thus, considering the Avogadro's number, we can write:
6.022 x 10^23 atoms Ti -------------------- 1 mol Ti
2.50 x 10^23 atoms Ti ---------------------- x
Solving for x, we'll have:
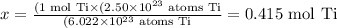
Therefore, 2.50 x 10^23 atoms of titanium correspond to 0.415 moles of titanium.