Answer:
The percent yield is 24.2%.
Step-by-step explanation:
1st) It is necessary to balance the chemical reaction:
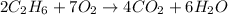
2nd) From the balanced reaction, we know that 2 moles of C2H6 react with 7 moles of O2 to produce 4 moles of CO2. With the molar mass of C2H6 (30g/mol), O2 (32g/mol) and CO2 (44g/mol), we can convert the moles to grams:
- C2H6 conversion:
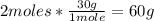
- O2 conversion:
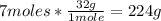
-CO2 conversion:
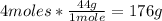
Now we know that 60g of C2H6 react with 224g of O2 to produce 176g of CO2.
3rd) From the given values of C2H6 (6.0g) and O2 (20.0g), it is necessary to find out which one is the limiting reactant and which one is the excess reactant:
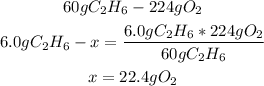
We can see that the 6.0g of C2H6 will need 22.4g of O2 to react, but we only have 20.0g of O2, so O2 is the limiting reactant and C2H6 will be the excess reactant.
4th) Now, using the limiting reactant, we have to calculate the grams of CO2 that should be produced from the stoichiometry of the reaction (this is the Theoretical yield):
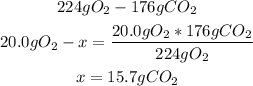
5th) Finally, we can calculate the Percent yield of the reaction, by using the Theoretical yield (15.7g) and the Actual yield (3.80g):
![\begin{gathered} PercentYield=(ActualYield)/(TheoreticalYield)*100\% \\ PercentY\imaginaryI eld=(3.80g)/(15.7g)*100\operatorname{\%} \\ PercentY\mathrm{i}eld=24.2\% \end{gathered}]()
So, the percent yield is 24.2%.