Answer:
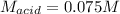
Step-by-step explanation:
Hello!
In this case, since the reaction between NaOH and HNO3 is:
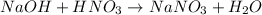
Whereas there is a 1:1 mole ratio between the acid and base, thus, we can write:
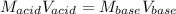
In such a way, solving for the concentration of the acid, HNO3, we obtain:
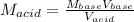
Therefore, by plugging in we obtain:
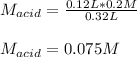
Best regards!