Answer: The mass of gold bar is 658.516 g
Step-by-step explanation:
To calculate the volume of cuboid, we use the equation:
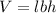
where,
V = volume of cuboid
l = length of cuboid = 4.21 cm
b = breadth of cuboid = 2.98 cm
h = height of cuboid = 2.72 cm
Putting values in above equation, we get:
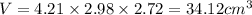
To calculate the mass of a substance, we use the equation:
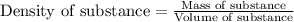
Density of gold bar =
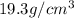
Volume of gold bar =
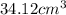
Putting values in equation 1, we get:
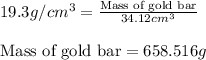
Hence, the mass of gold bar is 658.516 g