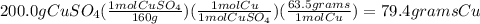1)we need a balanced equation: CuSO₄ + Zn ---> ZnSO₄ + Cu
2) we need to convert the grams of CuSO₄ to moles using the molar mass.
molar mass CuSO₄= 63.5 + 32.0 + (4 x 16.0)= 160 g/mol
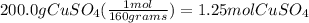
3) convert moles of CuSO₄ to moles of Cu
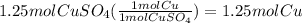
4) convert moles of Cu to grams using it's molar mass.
molar mass Cu= 63.5 g/mol
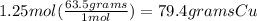
I did it step-by-step as the explanation but you can do all of this in one step.