ANSWER
The volume of oxygen gas at STP IS 23. 57152L
EXPLANATION;
Given that;
The mass of aluminum is 37.86g
To find the volume of oxygen at STP, follow the steps below
Step 1; Write a balanced equation for the reaction
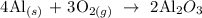
In the reaction above, 4 moles of Al reacts with 3 moles of O2
Step 2; Find the number of moles of Al using the formula below
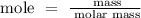
Recall, that the molar mass of Al is 26.98 g/mol
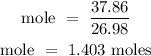
Therefore, the number of moles of Al is 1.403 moles
Step 3; Find the moles of O2 using a stoichiometry ratio
4 moles of Al is equivalent to 3 moles of oxygen
Let x represent the number of moles of oxygen
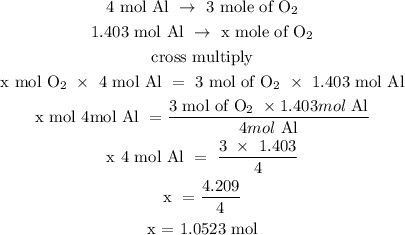
Therefore, the number of moles of oxygen is 1.0523 moles
Step 4; Find the volume of oxygen gas
1 mol of gas at S.T.P is 22.4
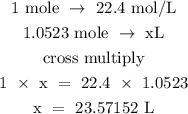
Hence, the volume of oxygen gas at STP IS 23. 57152L