Answer:
75 milligrams.
Explanation:
We have been given that Bismuth-210 has a half-life of about 5 days.
To solve our given problem we will use half-life formula.
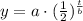 , where,
, where,
a = Initial value,
t = Time,
b = half-life.
Upon substituting our given values in above formula we will get,
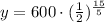
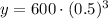
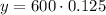
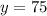
Therefore, 75 milligrams of Bismuth-210 will remain after 15 days.