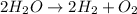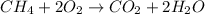They give us the reactions with their respective products and reactants. To balance a reaction we must bear in mind that matter is neither created nor destroyed, it only transforms. So the mass that we have in the reactants must be the same in the products. We verify this by counting the atoms of each element on each side of the reaction.
In the first reaction we have nitrogen and hydrogen. We have 2 nitrogens and 2 hydrogens in the reactants. We start off-balanced in nitrogen, so we place coefficient 2 in the NH3 molecule to get 2NH3. Now that we have 2 nitrogens and 6 hydrogens in the products, we must balance the hydrogen in the reactants. For that, we place the coefficient 3 in the H2 in such a way that there will be 6 hydrogen atoms in the reactants. The equation is balanced and will be:
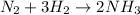
We do this same procedure for the other two equations. We count the atoms, we put the coefficients so that the number of atoms is conserved and we count again until the number of atoms of each element is the same on each side of the reaction. For the other two reactions we have: