Answer: The mass percent of NaCl solution is 16.66 %
Step-by-step explanation:
To calculate the mass percentage of NaCl solution, we use the equation:
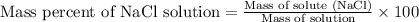
Mass of solute (NaCl) = 500.0 g
Mass of solvent (water) = 2.50 kg = 2500 g (Conversion factor: 1 kg = 1000 g)
Mass of solution = Mass of solute + Mass of solvent = 500.0 + 2500 = 3000 g
Putting values in above equation, we get:

Hence, the mass percent of NaCl solution is 16.66 %