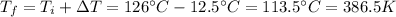Missing question:
"What is the final temperature of the gas?"
Solution:
The first law of thermodynamics states that:
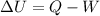
where
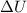
is the variation of internal energy of the gas
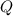
is the heat absorbed by the gas
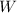
is the work done by the gas
The gas in this problems absorbs
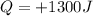
of heat and it does
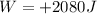
of work, so its variation of internal energy is:
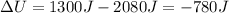
This means the gas has lost internal energy.
But the variation of internal energy is related to the variation of temperature by:
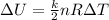
where
k is the number of degrees of freedom (k=3 for a monoatomic gas)
n is the number of moles
R is the gas constant

is the variation of temperature of the gas.
Re-arranging the equation and using the variation of internal energy that we found at the previous step, we find:
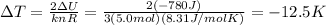
Variation of temperatures in Kelvin are equal to variation of temperatures in Celsius, so
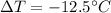
and we can now find the final temperature of the gas: