Answer:
S₄O₁Cl₈
Step-by-step explanation:
Given,
Mass of S = 26.96 g
Mass of O = 13.45 g
Mass of Cl = 59.59 g
Atomic mass of S = 32.065 u
Atomic mass of O = 15.999 u
Atomic mass of Cl = 35.453 u
Moles of S =
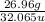 = 0.84
= 0.84
Moles of O =
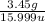 = 0.2156
= 0.2156
Moles of Cl =
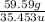 = 1.68
= 1.68
Dividing by the smallest mole to get molar ratio:
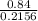 ,
,
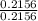 ,
,
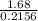
3.89, 1, 7.79
~ 4, 1, 8
The empirical formula is S₄O₁Cl₈