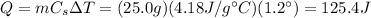The increase in temperature of the water is
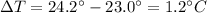
The total mass of the water is the product between its volume and its density
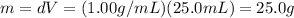
And so we can find the amount of heat released by the pebble to the water, because this is equal to the amount of heat absorbed by the water, which is