Step-by-step explanation:
An isotope is defined as the specie which contains same number of protons but different number of neutrons.
Also, it is known that atomic number is the sum of only total number of protons present in an element. Whereas mass number is the sum of total number of both protons and neutrons present in an element.
Hence, atomic mass of the given atom will be as follows.
Atomic mass = no. of protons + no. of neutrons
= 77 + 117
= 194
According to the periodic table, platinum is the atom with atomic number 194.
Hence, we can conclude that isotope that has atoms with 117 neutrons 77 protons and 77 electrons is
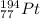 .
.