Answer: The volume of liquid is 16.2 mL
Step-by-step explanation:
To calculate volume of a substance, we use the equation:
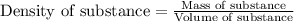
We are given:
Density of liquid = 0.789 g/mL
Mass of liquid = 12.8 g
Putting values in above equation, we get:
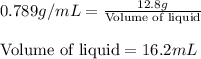
Hence, the volume of liquid is 16.2 mL