Answer:
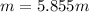
Step-by-step explanation:
Hello,
Molality is defined as:
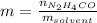
Thus, as the solution is % by mass, one assumes that the mass of urea is 26g and the mass of the solution 100g, in such a way, one computes the moles of urea by knowing its molecular mass:
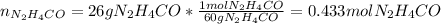
Now, as solution has a mass of 100g, the solvent has a mass of 74g which corresponds to 0.074L since the density of water is 1g/mL; in such a way the molality is:
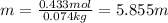
Best regards.