Answer:
111 L
Step-by-step explanation:
Calculation of moles of hydrogen gas:-
Mass of
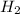 = 18.6 g
= 18.6 g
Molar mass of
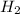 = 2.01588 g/mol
= 2.01588 g/mol
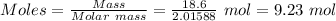
According to the given reaction:-
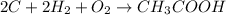
2 moles of hydrogen gas on reaction produces one mole of acetic acid gas.
So,
1 mole of hydrogen gas on reaction produces
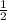 mole of acetic acid gas.
mole of acetic acid gas.
Also,
9.23 mole of hydrogen gas on reaction produces
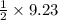 mole of acetic acid gas.
mole of acetic acid gas.
Moles of acetic acid gas = 4.615 moles
Given that:
Temperature = 35 °C
The conversion of T( °C) to T(K) is shown below:
T(K) = T( °C) + 273.15
So,
T = (35 + 273.15) K = 308.15 K
n = 4.615 moles
P = 1.05 atm
V = ?
Using ideal gas equation as:
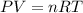
where,
P is the pressure
V is the volume
n is the number of moles
T is the temperature
R is Gas constant having value = 0.0821 L atm/ K mol
Applying the equation as:
1.05 atm × V = 4.615 moles ×0.0821 L atm/ K mol × 308.15 K
⇒V = 111 L