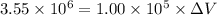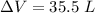Answer:
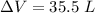
Step-by-step explanation:
According to the first law of thermodynamics:-
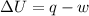
Where,
U is the internal energy =
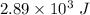
q is the heat =
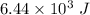 (heat is gained)
(heat is gained)
w is the work done = ?
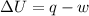
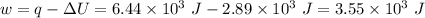
The expression for the calculation of work done is shown below as:
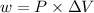
Where, P is the pressure,
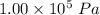
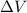 is the change in volume
is the change in volume
Also, 1 J = 1000 PaL
So,

From the question,