Answer:
B. Pb
Step-by-step explanation:
I have used yellow dots to mark your elements on the Periodic Table below.
Their melting points are
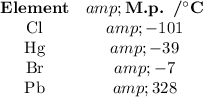
The Periodic Table and the list of melting points both show that lead is the only element in at your list that is a solid at STP.