Answer:
m = 40.88 g
Step-by-step explanation:
Given data:
We know that Ice at zero degree celcius convert into liquid form by observing heat
latent heat of fusion = 79.5 cal/g
heat required or relaesed to convert 50 gram of water at 65 degree c to zero degree celcius
let mass of ice is m
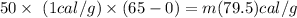
where 1 cal/g os specific heat of water
solving for m
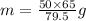
m = 40.88 g