Answer:
Explained
Step-by-step explanation:
Heat dissipated is Q=
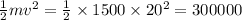 J
J
Temperature in Kelvin is 273+200=293 K
So
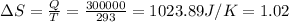 kJ/K
kJ/K
b. Entropy is the measurement randomness of a system. It increases with increase in reversibility.
c. Heat generated is due to friction(Q) and if we could reduce this friction heat to zero, we can cause the system have zero change in entropy. This can be done with help of proper lubrication. Lubrication can make the system adiabatic.