Answer: a) the numbered energy levels starting from the nucleus outward
Step-by-step explanation:
Principle Quantum Number: It describes the size of the orbital and the main energy level. It is represented by n. Where, n = 1,2,3,4....stands for K, L, M, N shell and so on..
Azimuthal Quantum Number : It describes the shape of the orbital. It is represented as 'l'. The value of l ranges from 0 to (n-1). For l = 0,1,2,3... the orbitals are s, p, d, f...
Magnetic Quantum Number : It describes the orientation of the orbitals. It is represented as
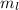 . The value of this quantum number ranges from
. The value of this quantum number ranges from
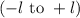 . When l = 2, the value of
. When l = 2, the value of
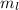 will be -2, -1, 0, +1, +2.
will be -2, -1, 0, +1, +2.
Spin Quantum number : It describes the direction of electron spin. This is represented as
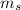 The value of this is
The value of this is
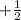 for upward spin and
for upward spin and
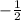 for downward spin.
for downward spin.