Answer:
An atom accepts electrons and becomes an anion.
Step-by-step explanation:
Atomic number = number of electrons = number of protons.
Atomic number of sodium is 11
So the atom contains 11 protons and 11 electrons
To find the number of neutrons we make use of the formula
Mass number - atomic number = number of neutrons
From the periodic table, we know mass number of sodium is 23
So number of neutron = 23 - 11 = 12.
When a sodium atom loses an electron it will have 11 positive protons and 10 negative electrons. Since 1 positive charge is more, Na becomes
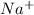 .
.
Positively charged ion is called as cation
Chlorine's atomic number is 17 so it has 17 protons and 17 electrons.
When it gains an electrons, it will have 17 positive protons and 18 negative electrons. Since 1 negative charge is more, Cl becomes
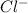 .
.
Negatively charged ion is called as anion.