Answer: increase in entropy
Step-by-step explanation:
Entropy is the measure of randomness or disorder of a system. If a system moves from an ordered arrangement to a disordered arrangement, the entropy is said to decrease and vice versa.
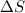 is positive when randomness increases and
is positive when randomness increases and
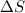 is negative when randomness decreases.
is negative when randomness decreases.
When the non polar solutes are placed in water, the hydrogen bonding network of water is disrupted, and there are fewer ways for water to hydrogen-bond with itself. That means the water molecules are more randomly arranged and thus have more entropy and thus
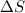 is positive.
is positive.