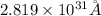Answer : The length of a one-dimensional box for an electron is
Explanation :
The energy level of quantum particle in a one-dimensional box is given as:
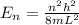
where,
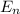 = 479 kJ/mol = 479000 J/mol
= 479 kJ/mol = 479000 J/mol
n = energy level = 1
h = Planck's constant =
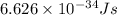
m = mass of electron =
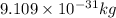
L = length of a one-dimensional box = ?
Now put all the given values in the above formula, we get:
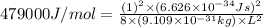
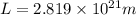
conversion used :
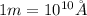
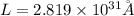
Therefore, the length of a one-dimensional box for an electron is