Answer:
molar volume of vapor ethanol is
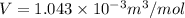
mass of ethanol is 15430.22 g
Step-by-step explanation:
By using ideal gas equation
PV = nRT
Where P is pressure , R is gas constant
so, volume is
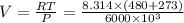
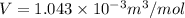
molar volume of vapor ethanol is
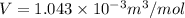
b)
volume of vessel is given
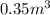
therefore total moles of ethanol in given vessel will be
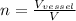
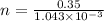
n = 335.44 mol
we know that
mole is given as [/tex]n = \frac{mass}{moleculae weight}[/tex]
weight of ethanol is 46 g/mol
n\times 46 = mass
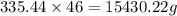
mass of ethanol is 15430.22 g