Answer:
Time to decay will be 377.7 days.
Explanation:
Decay of an radioactive element is represented by the formula
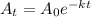
where
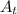 = Amount after t days
= Amount after t days
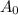 = Initial amount
= Initial amount
t = duration for the decay
k = decay constant
Now we plug in the values in the formula
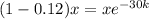
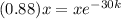
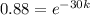
Now we take natural log on both the sides
ln(0.88) =
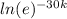
ln(0.88) = -30k(lne)
-30k = -0.1278
k =
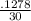
k =
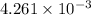
Now we have to calculate the duration for the decay of 50 mg sample to 10 mg.
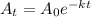
We plug in the values in the formula
10 = 50
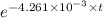
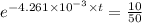
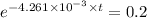
We take (ln) on both the sides
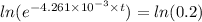
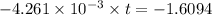
t =
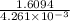
t = 0.37771×10³
t = 377.7 days
Therefore, time for decay will be 377.7 days.