Answer: The percent yield of the given reaction is 82 %.
Step-by-step explanation:
The chemical equation for the reaction of zinc and hydrochloric acid follows:
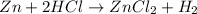
To calculate the percent yield of the reaction, we use the equation:
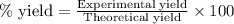
Experimental yield of hydrogen gas = 164 g
Theoretical yield of hydrogen gas = 200 g
Putting values in above equation, we get:
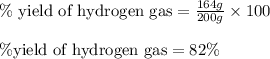
Hence, the percent yield of the reaction is 82 %.