Answer: The most likely identity of the substance is iron.
Step-by-step explanation:
The quantity of heat required to raise the temperature of a substance by one degree Celsius is called the specific heat capacity.
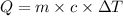
Q = Heat absorbed=
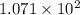 Joules
Joules
m= mass of substance = 11.9 g
c = specific heat capacity = ?
Initial temperature of the water =
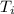 = 25.0°C
= 25.0°C
Final temperature of the water =
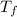 = 45.0°CChange in temperature ,
= 45.0°CChange in temperature ,
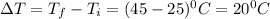
Putting in the values, we get:
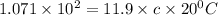
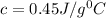
The specific heat of 0.45 is for iron and thus the substance is iron.