Answer : The molarity of a formic acid solution is, 0.0903 M
Explanation :
To calculate the molarity of formic acid, we use the equation given by neutralization reaction:
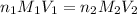
where,
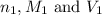 are the n-factor, molarity and volume of formic acid which is
are the n-factor, molarity and volume of formic acid which is
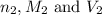 are the n-factor, molarity and volume of sodium hydroxide base which is NaOH.
are the n-factor, molarity and volume of sodium hydroxide base which is NaOH.
As we are given:
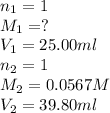
Now put all the given values in above equation, we get:
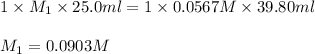
Hence, the molarity of a formic acid solution is, 0.0903 M