Answer:
60.00% is the volume percent the ethylene glycol in the antifreeze mixture.
Step-by-step explanation:
Volume of the ethylene glycol in the mixture = 3.00 gal
Volume of the water in the mixture = 2.00 gal
Total volume of the mixture =3.00 gal + 2.00 gal = 5.00 gal
Volume percent the ethylene glycol :
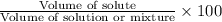
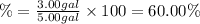
60.00% is the volume percent the ethylene glycol in the antifreeze mixture.