Step-by-step explanation:
The law of conservation of matter states that the in chemical reaction the total mass on the left side of the equation should be equal to the total mass on the right side of the equation i.e. total mass on the both sides of the equation must be equal.
Option (1)
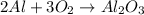
Molar mass of reactant -
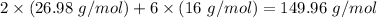
Molar mass of product -
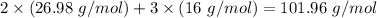
LHS ≠ RHS
Law of conservation of matter not followed
Option (2)
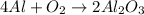
Molar mass of reactant -
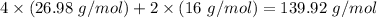
Molar mass of product -
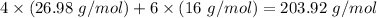
The law of conservation of matter is not followed.
Option (3)
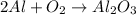
Molar mass of reactant -
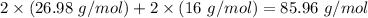
Molar mass of product -
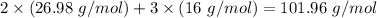
The law of conservation of matter is not followed.
Option (4)
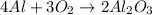
Molar mass of reactant -
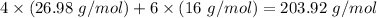
Molar mass of product -
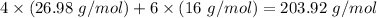
LHS = RHS
The law of conservation of matter is followed. Hence, the correct option is (D).